
Bernard Ribalet, PhD
Position Title:Research Interests:
For a number of years my main goal has been to study the structure and function of inward rectifier K channels, using the patch clamp technique together with mutagenesis. In particular, we have characterized the residues that interact with PIP2 to activate Kir2.1 and Kir6.2. The latter interacts with SUR1 to form one of the ATP-dependent K channels. We have also characterized the Kir6.2 residues that bind ATP and modeled how this interaction alters channel gating. In addition to studying the regulation of channel gating by PIP2 and ATP, we have investigated the trafficking of wild-type and mutant inward rectifier K channels by linking the pore forming protein to the green fluorescent protein, GFP.
More recently, I have focused on the glucose homeostasis in skeletal and cardiac muscle cells. Insulin-dependent glucose uptake in skeletal muscle is essential to lower blood glucose and prevent diabetes. In cardiac muscle glucose and insulin are cardioprotective. We have used a probe that undergoes changes in FRET upon binding and unbinding intracellular glucose to assess how some ligands and genes affect glucose uptake as well as glucose utilization and glycogen synthesis in these tissues.
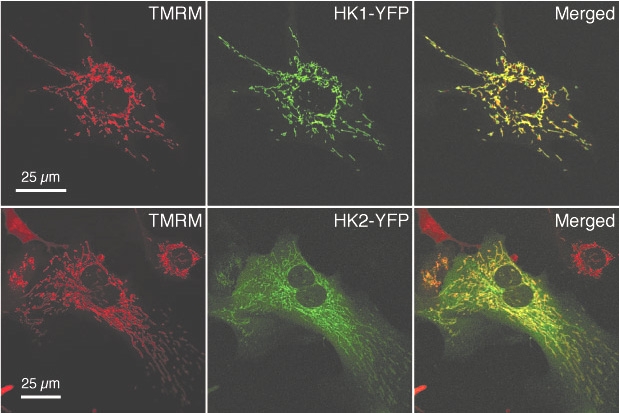
Differential Localization of HKI-YFP and HKII-YFP in Neonatal Cardiac Myocytes.
Representative Publications:
John S.A, Weiss J.N and Ribalet B. (2011). Subcellular localization of hexokinases I and II directs the metabolic fate of glucose. PLoS One. Mar 9;6(3):e17674.
Ribalet, B., John, S. A., Xie, L. H. and Weiss, J. N. (2006) ATP-sensitive K+ channels: regulation of bursting by the sulphonylurea receptor, PIP2 and regions of Kir6.2. Journal of Physiology. 571, 303-17.
John S.A., Weiss J.N., Xie L-H., and Ribalet B. (2003) Molecular mechanism for ATP-dependent closure of the K+ channel Kir6.2. Journal of Physiology. 552:23-34.